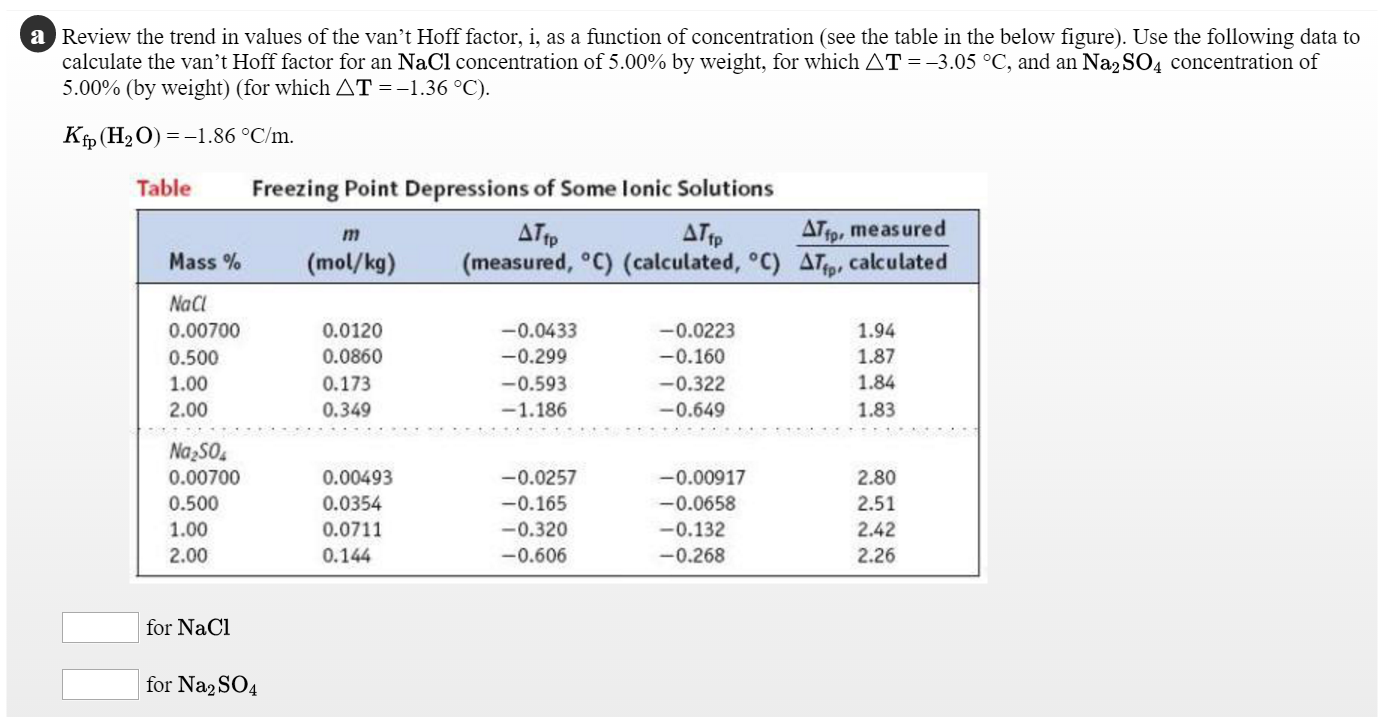
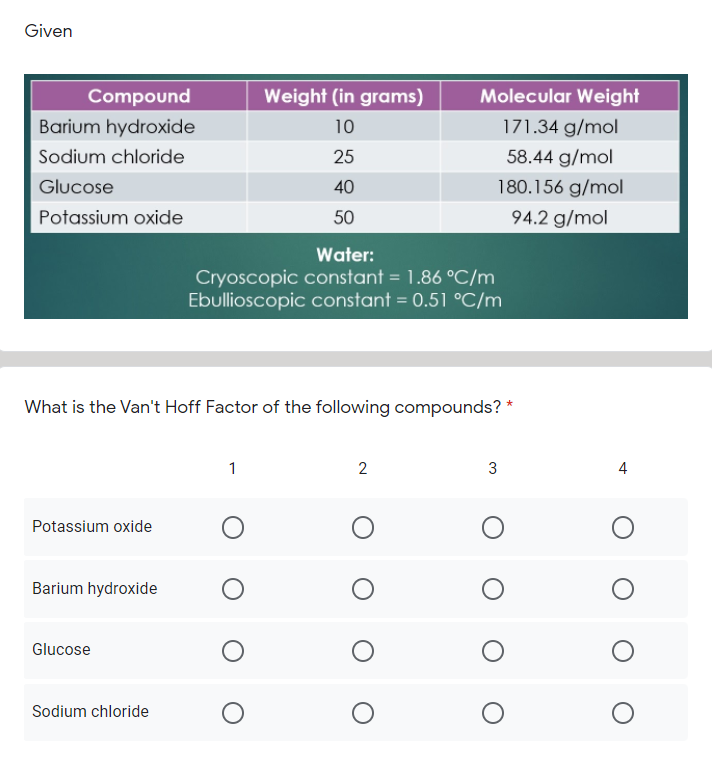
SOLVED: Question 2 0.5 pts What is the van't Hoff factor for methanol? Question 3 0.5 pts What is the van't Hoff factor for ethanol?

Relationship of COP and van't Hoff factor, i for aqueous salt solutions | Download Scientific Diagram

Predicting Van't Hoff (i) Factors: Colligative Properties | Chemistry worksheets, Teaching chemistry, Chemistry
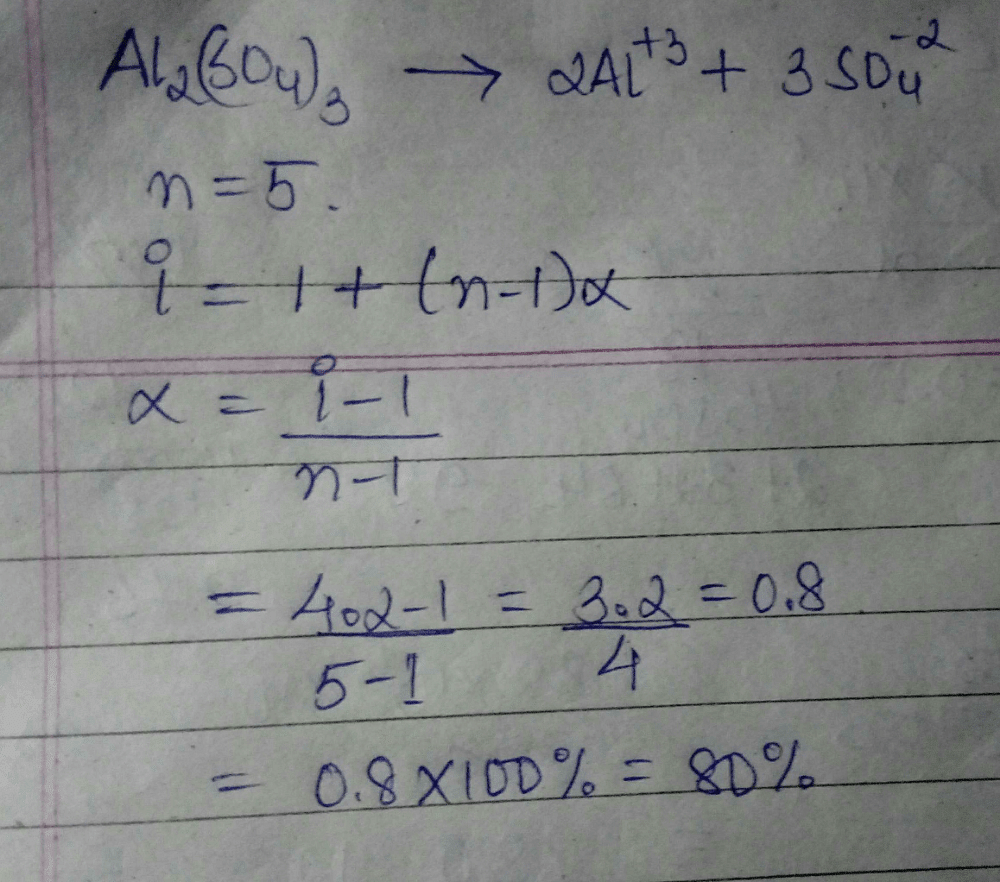
The van't Hoff factor for a 0.1 M solution is4.20. The degree of dissociation isa)80%b)90%c)78%d)83%Correct answer is option 'A'. Can you explain this answer? | EduRev NEET Question

Van't Hoff factor as a function of concentration (calculated according... | Download Scientific Diagram